Asia Cervical Cancer Diagnostics Market Size and Forecast 2026–2034
Asia Cervical Cancer Diagnostics Market Size and Forecast 2026–2034
Introduction
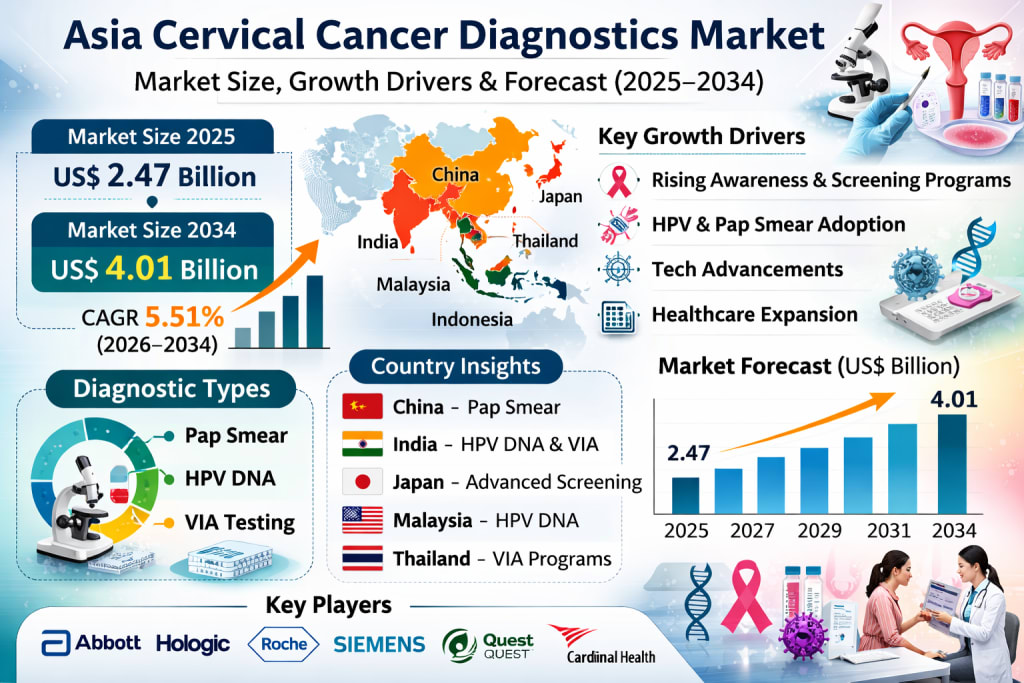
The Asia cervical cancer diagnostics market is entering a critical phase of growth, driven by rising awareness, improving healthcare access, and the rapid adoption of advanced diagnostic technologies. According to Renub Research, the market is anticipated to grow from US$ 2.47 billion in 2025 to US$ 4.01 billion in 2034, registering a compound annual growth rate (CAGR) of 5.51% during the forecast period from 2026 to 2034. This steady expansion reflects a broader regional shift toward preventive healthcare, early disease detection, and stronger public health systems focused on women’s health.
Cervical cancer remains one of the most preventable yet still deadly cancers affecting women, particularly in low- and middle-income countries across Asia. The disease is highly treatable when detected early, but late-stage diagnosis continues to be a major challenge in several parts of the region. As a result, governments, healthcare institutions, and non-governmental organizations are increasingly prioritizing screening programs and awareness campaigns to reduce mortality rates and long-term healthcare costs.
Asia Cervical Cancer Diagnostics Market Outlook
Cervical cancer diagnostics refer to the range of medical tests and procedures used to detect cervical cancer and its precancerous conditions. These include Pap smear tests, human papillomavirus (HPV) tests, colposcopy, biopsy, and other advanced diagnostic technologies. Early diagnosis plays a crucial role in improving survival rates, as treatment is significantly more effective when the disease is identified at an early stage.
In Asia, the adoption of cervical cancer diagnostics has been steadily increasing due to a combination of factors: growing awareness of women’s health, rising incidence of cervical cancer, and the expansion of healthcare infrastructure. Historically, many Asian countries struggled with limited screening coverage, leading to a high proportion of late-stage diagnoses. Today, however, national screening programs, HPV vaccination drives, and public health campaigns are reshaping the landscape.
The region’s large and diverse population presents both a challenge and an opportunity. While disparities in access remain, improvements in diagnostic capacity, the spread of cost-effective testing methods, and the involvement of private healthcare providers are making cervical cancer screening more accessible than ever before. With continued policy support and technological progress, the Asia cervical cancer diagnostics market is expected to maintain strong momentum throughout the forecast period.
Growth Drivers of the Asia Cervical Cancer Diagnostics Market
Rising Awareness and Government-Led Screening Programs
One of the most powerful drivers of market growth is the increasing awareness of cervical cancer and the expansion of government-led screening initiatives. Many Asian countries have launched national programs aimed at promoting early detection through Pap smear tests, HPV testing, and other affordable screening methods, including outreach in rural and underserved areas.
The primary goal of these programs is to educate women about the importance of regular screening and early diagnosis. As awareness improves, participation rates in screening programs are rising, particularly among younger and more health-conscious populations. This shift is helping to normalize preventive gynecological check-ups and reduce the stigma historically associated with reproductive health issues.
In September 2024, the United States, Australia, India, and Japan announced a landmark initiative under the Quad Leaders Summit to help end cancer as it is currently known in the Indo-Pacific, starting with cervical cancer. The Quad Cancer Moonshot aims to strengthen health infrastructure, expand research partnerships, develop data systems, and enhance support for cancer prevention, detection, treatment, and care. Such international collaborations are expected to further accelerate the adoption of cervical cancer diagnostics across Asia.
Technological Advancements in Diagnostic Methods
Technological innovation is another major force propelling the market forward. The development of highly sensitive HPV testing, molecular diagnostics, liquid-based cytology, and automated screening platforms has significantly improved the accuracy and efficiency of cervical cancer screening.
These advanced tools enable earlier detection of precancerous lesions and high-risk HPV strains, allowing timely medical intervention. Automation and digital imaging also reduce the risk of human error and increase screening throughput—an essential advantage in densely populated Asian countries with large screening needs.
Point-of-care testing and self-sampling solutions are further expanding access, particularly in remote and resource-limited areas. As production scales up and costs decline, these modern diagnostic technologies are becoming more affordable and widely available.
In September 2025, PHASE Scientific International Limited announced the launch of the world’s largest clinical study for urine-based cervical cancer (HPV) screening, with the first site established in Guang’an, Sichuan Province, China. This milestone highlights how innovation is reshaping the future of cervical cancer diagnostics and making screening more convenient and accessible.
Improving Healthcare Infrastructure and Access
The steady improvement of healthcare infrastructure across Asia is also supporting market growth. Many countries are investing heavily in hospitals, diagnostic centers, and primary healthcare facilities to strengthen their diagnostic capabilities. The rise of private diagnostic chains and partnerships with global healthcare organizations has further improved service availability and quality.
Urbanization and higher healthcare spending have increased the number of skilled medical professionals and advanced laboratories. At the same time, mobile screening units and community-based healthcare models are helping extend services to underserved populations.
In January 2025, the Asian Infrastructure Investment Bank (AIIB) introduced its first-ever Health Strategy, signaling a strong commitment to social development and inclusive healthcare. Such initiatives are expected to improve access to screening services and further boost the cervical cancer diagnostics market in the region.
Challenges Facing the Asia Cervical Cancer Diagnostics Market
Limited Awareness and Cultural Barriers
Despite progress, limited awareness and cultural barriers remain significant obstacles in several Asian countries. In conservative or rural communities, social stigma, fear, and misconceptions about cervical cancer and screening procedures can discourage women from seeking regular check-ups. Embarrassment, lack of education, and misinformation often lead to delayed or avoided screenings, increasing the risk of late-stage diagnosis.
Uneven Access and Resource Constraints
Another major challenge is the uneven distribution of healthcare resources. While urban areas often have access to advanced diagnostic facilities, rural and low-income regions may lack skilled personnel, laboratories, and modern equipment. The high cost of advanced diagnostics can also limit adoption, especially in countries with constrained public healthcare budgets and inconsistent reimbursement systems.
Market Segmentation Analysis
Asia Cervical Cancer Pap Smear Diagnostics Market
Pap smear testing remains a cornerstone of cervical cancer screening across Asia. It is widely used to detect abnormal cervical cells at an early stage and is valued for its cost-effectiveness and established clinical acceptance. In many countries, Pap smear tests are part of routine public health programs and gynecological check-ups.
Although newer technologies are gaining ground, Pap smear diagnostics continue to play a critical role, especially in large-scale screening initiatives. The adoption of liquid-based cytology in urban healthcare settings has further improved test accuracy and efficiency, supporting stable demand for this segment.
Asia Cervical Cancer HPV DNA Diagnostics Market
The HPV DNA diagnostics segment is expanding rapidly due to its high sensitivity and ability to detect high-risk HPV strains before cellular abnormalities appear. Compared to Pap tests, HPV DNA testing allows for longer screening intervals and earlier risk identification.
Technological improvements and declining costs are making these tests more accessible, and growing awareness of the link between HPV and cervical cancer is accelerating adoption. This segment is expected to be one of the fastest-growing areas of the Asia cervical cancer diagnostics market.
Asia Cervical Cancer VIA Diagnostics Market
Visual Inspection with Acetic Acid (VIA) is widely used in resource-poor and rural areas because of its simplicity, low cost, and immediate results. VIA does not require sophisticated laboratory infrastructure, making it ideal for community-based screening programs and large-scale public health initiatives.
Although less specific than molecular tests, VIA remains a vital tool for expanding screening coverage in underserved regions. Its continued use will be essential for reducing cervical cancer mortality where access to advanced diagnostics is limited.
Country-Level Market Insights
China Cervical Cancer Pap Smear Market
China’s Pap smear market is supported by large public health programs and a growing emphasis on preventive care. With its massive female population, increasing awareness, and improving laboratory infrastructure, Pap smear testing remains a key screening tool, particularly in urban hospitals and maternal health centers. While HPV testing is gaining popularity, Pap smears continue to be a cost-effective and widely used option.
Japan Cervical Cancer Pap Smear Market
Japan has a mature and technologically advanced Pap smear market, driven by strong healthcare standards and a culture of regular health check-ups. Automated screening systems, skilled professionals, and high-quality laboratories support consistent demand. Even as HPV testing grows, Pap smears remain a central part of Japan’s cervical cancer screening strategy.
India Cervical Cancer HPV DNA Market
India’s HPV DNA market is growing rapidly due to rising awareness, expanding private healthcare, and updated screening guidelines. Although cost remains a concern, declining prices and increasing availability are improving access. Government and NGO initiatives are also supporting the adoption of advanced screening technologies.
Malaysia Cervical Cancer HPV DNA Market
Malaysia is seeing steady growth in HPV DNA testing, particularly in private hospitals and diagnostic centers. Government-backed programs, improved laboratory capacity, and a strong focus on preventive healthcare are driving adoption among urban and insured populations.
Thailand Cervical Cancer VIA Market
In Thailand, VIA plays a crucial role in expanding screening access, especially in rural and low-resource settings. Its low cost and ease of use make it ideal for community health programs aimed at reducing cervical cancer mortality.
India Cervical Cancer VIA Market
VIA remains a vital screening method in India’s rural and underserved regions. Its affordability and immediate results make it suitable for large-scale public health initiatives, helping bridge the gap where laboratory-based testing is not readily available.
Market Segments
By Type:
Pap Smear Market
HPV DNA Market
VIA (Visual Inspection with Acetic Acid) Market
Pap Smear: Japan, Korea, Singapore, Malaysia, India, China, Thailand, Indonesia
HPV DNA: Japan, Korea, Singapore, Malaysia, India, China, Indonesia
VIA: India, China, Thailand, Indonesia
Competitive Landscape
Companies are analyzed across five viewpoints: Overview, Key Persons, Recent Developments & Strategies, Product Portfolio, and Financial Insights.
Key Companies Covered:
Abbott Laboratories
Hologic Corporation
Becton
Siemens AG
Roche Diagnostics
Quest Diagnostics
Cardinal Health
These players are focusing on innovation, partnerships, and geographic expansion to strengthen their presence in the fast-growing Asian market.
Final Thoughts
The Asia cervical cancer diagnostics market is on a strong growth trajectory, supported by rising awareness, government initiatives, technological innovation, and improving healthcare infrastructure. With the market projected to grow from US$ 2.47 billion in 2025 to US$ 4.01 billion by 2034 at a CAGR of 5.51%, the region is clearly moving toward a future where early detection and prevention play a central role in women’s healthcare.
While challenges related to awareness, access, and affordability remain, continued investment, policy support, and innovation are expected to narrow these gaps. As screening becomes more accessible and socially accepted, cervical cancer diagnostics will not only represent a growing market opportunity but also a critical pillar in improving public health outcomes across Asia.






Comments
There are no comments for this story
Be the first to respond and start the conversation.